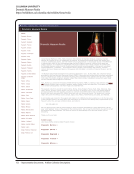
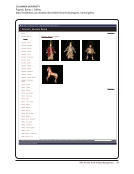
SPEC Kit 333: Art &Artifact Management · 81
National Library of Medicine
Collection Development Policy. Images and Archives Section
Collection Development Policy—Images and Archives -3 -
The Images and Archives Collections will not accept the following:
e. Collections where Library activities (e.g., of preservation) will assist in
underwriting the donor’s earning royalties or otherwise enable the
donor to profit. (However, if the simple fact of NLM’s acquisition or
exhibition of collection materials happens to profits the donor, that
in itself becomes no bar to acquisition, as long as exhibition is not a
requirement of donation.)
f. Collections that in whole or in part may not be deaccessioned at the
Library’s discretion.
Subject Matter Focuses for the Images and Archives Collections
I anticipate actively developing the collections in five broad areas that help tell the story of
twentieth- and twenty-first–century health and medicine.
1. Biomedical Research: papers of leading researchers/research
administrators/professional societies.
Focus on those who researchers who have won the Nobel prize or the Lasker award,
or who have risen to prominence within the National Institutes of Health.
Focus areas, building to the strengths of the collections:
a. Molecular medicine and cellular regulation: neural and hormonal action/
signal transduction (Axelrod, Rodbell) genetics and genetic engineering
(Fredrickson, Lederberg, Cohen).
b. Human psychosocial development (child development papers, Bowen).
c. International health, especially tropical health (Soper, Work).
d. Medical informatics (Collen, American Association for Medical Systems and
Informatics, NLM Archives). Represents a library focus.
e. Health Services Research (Eisenberg). Represents a library focus.
2. Biotechnology/Drugs.
Relying on traditional strengths of the collection and active connections to possible
future areas, as well as the possibility for exhibition
a. Artificial Internal Organs/transplantation (ASAIO Project).
b. Alternative and Non-Traditional Devices/Fraud/Quackery/ (FDA Case
files, advertising materials)
Acquiring the papers of biotechnology firms is important, as these corporate
archives are likely not to survive if the firm goes out of business. Though it’s not
been a traditional focus, I’d like also to consider:
National Library of Medicine
Collection Development Policy. Images and Archives Section
Collection Development Policy—Images and Archives -3 -
The Images and Archives Collections will not accept the following:
e. Collections where Library activities (e.g., of preservation) will assist in
underwriting the donor’s earning royalties or otherwise enable the
donor to profit. (However, if the simple fact of NLM’s acquisition or
exhibition of collection materials happens to profits the donor, that
in itself becomes no bar to acquisition, as long as exhibition is not a
requirement of donation.)
f. Collections that in whole or in part may not be deaccessioned at the
Library’s discretion.
Subject Matter Focuses for the Images and Archives Collections
I anticipate actively developing the collections in five broad areas that help tell the story of
twentieth- and twenty-first–century health and medicine.
1. Biomedical Research: papers of leading researchers/research
administrators/professional societies.
Focus on those who researchers who have won the Nobel prize or the Lasker award,
or who have risen to prominence within the National Institutes of Health.
Focus areas, building to the strengths of the collections:
a. Molecular medicine and cellular regulation: neural and hormonal action/
signal transduction (Axelrod, Rodbell) genetics and genetic engineering
(Fredrickson, Lederberg, Cohen).
b. Human psychosocial development (child development papers, Bowen).
c. International health, especially tropical health (Soper, Work).
d. Medical informatics (Collen, American Association for Medical Systems and
Informatics, NLM Archives). Represents a library focus.
e. Health Services Research (Eisenberg). Represents a library focus.
2. Biotechnology/Drugs.
Relying on traditional strengths of the collection and active connections to possible
future areas, as well as the possibility for exhibition
a. Artificial Internal Organs/transplantation (ASAIO Project).
b. Alternative and Non-Traditional Devices/Fraud/Quackery/ (FDA Case
files, advertising materials)
Acquiring the papers of biotechnology firms is important, as these corporate
archives are likely not to survive if the firm goes out of business. Though it’s not
been a traditional focus, I’d like also to consider: