SPEC Kit 336: Responsible Conduct of Research Training · 55
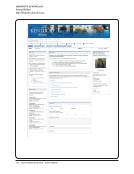
UNIVERSITY OF VIRGINIA
School of Medicine. Compliance and Training
http://www.medicine.virginia.edu/research/offices/research/home/compliance-training
Compliance and Training — School of Medicine at the University of Virginia
http://www.medicine.virginia.edu/research/offices/research/home/compliance-training[8/19/13 4:02:22 PM]
Patient Care Medical Center Physicians Group UVA HEALTH SYSTEM Claude Moore Library School of Nursing School of Medicine
Education Research Departments Community
SOM Home Research Research Home Compliance and Training
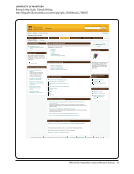
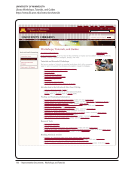
Compliance and Training
Summary and Quick links
Compliance
Research involving human subjects, animals, recombinant DNA/pathogens, biohazardous materials
Responsible conduct of research
Reporting misconduct
Conflict of interest and commitment
Current SOM requirements for the disclosure of external financial interests
Confidentiality
Clinical study-specific documentation
Faculty consulting agreements -criteria for School of Medicine review
Publication of clinical trials to web sites
Record retention and disposition
Terms and conditions of your award
Federal acts, circulars, and regulations pertaining to grants and contracts
NIH public access policy: School of Medicine site/Health Sciences Library site
Training
Office of Environmental Health and Safety: EHS home page /EHS training programs
Institutional Biosafety Committee
Animal Care and Use Committee
Institutional Review Boards
Other required training
SOM overview of safety issues associated with research areas (a "best practices" document)
Compliance
Human subjects research. The University has two Institutional Review Boards (IRBs). The IRB for the Social and
Behavioral Sciences (IRB-SBS) reviews and oversees non-medical, behavioral research studies. The IRB for Health
Sciences Research (IRB-HSR) oversees all other studies involving human subjects, representing the majority of
human use protocols performed by the School of Medicine. Investigators who are new to clinical research or who
wish to perform unfamiliar studies should contact the IRB-HSR prior to submitting a protocol. The Clinical Trials
Office can facilitate the conduct of clinical studies by assisting with budget and proposal preparation, study
coordination and management, and regulatory functions such as quality assurance/quality control.
Animals in research. The Institutional Animal Care and Use Committee (IACUC) reviews and oversees the use of
animals in research and teaching at the University. The IACUC provides training in handling research animals,
insures that individuals using research animals participate in the occupational health and safety program, and
conducts inspections of animal use facilities. The IACUC has specific protocol submission deadlines. The Center for
Comparative Medicine operates UVA vivaria and provides veterinary support. Current per diem rates are listed on
the CCM web site.
Recombinant DNA and pathogens. The Institutional Biosafety Committee (IBC) oversees the use of recombinant
DNA, organisms requiring at least Biosafety Level 2 conditions, and of human specimens. The IBC also inspects
laboratories that have registered for one or more of these activities.
Biohazardous materials (bloodborne pathogens, radiation, hazardous chemicals, shipping biological materials,
etc.). The Office of Environmental Health and Safety maintains comprehensive programs for the management of
potential hazards that may be encountered during research activities. Their web site includes current health and
safety policies and information on ordering radioactive materials. Click here for UVA training requirements,
programs, and on-line training.
Research
General information
Emergency information
Other offices supporting
research
Research resources and
collaborators
Research core facilities
For new faculty
For administrators
For students and postdocs
Intellectual property and
entrepreneurial activities
SOM review of proposed
consulting agreements
Forms and documents
Developing and submitting
a proposal
Where's that e-system?
FAQs
Links
Contact us
Home
Instructions for completing
CITI Conflict of Interest
Modules
Compliance and Training
Medicine PeoplePeople Search site U.Va. U.Va.
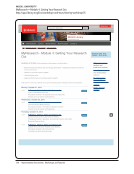
UNIVERSITY OF VIRGINIA
School of Medicine. Compliance and Training
http://www.medicine.virginia.edu/research/offices/research/home/compliance-training
Compliance and Training — School of Medicine at the University of Virginia
http://www.medicine.virginia.edu/research/offices/research/home/compliance-training[8/19/13 4:02:22 PM]
Patient Care Medical Center Physicians Group UVA HEALTH SYSTEM Claude Moore Library School of Nursing School of Medicine
Education Research Departments Community
SOM Home Research Research Home Compliance and Training
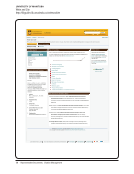
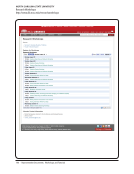
Compliance and Training
Summary and Quick links
Compliance
Research involving human subjects, animals, recombinant DNA/pathogens, biohazardous materials
Responsible conduct of research
Reporting misconduct
Conflict of interest and commitment
Current SOM requirements for the disclosure of external financial interests
Confidentiality
Clinical study-specific documentation
Faculty consulting agreements -criteria for School of Medicine review
Publication of clinical trials to web sites
Record retention and disposition
Terms and conditions of your award
Federal acts, circulars, and regulations pertaining to grants and contracts
NIH public access policy: School of Medicine site/Health Sciences Library site
Training
Office of Environmental Health and Safety: EHS home page /EHS training programs
Institutional Biosafety Committee
Animal Care and Use Committee
Institutional Review Boards
Other required training
SOM overview of safety issues associated with research areas (a "best practices" document)
Compliance
Human subjects research. The University has two Institutional Review Boards (IRBs). The IRB for the Social and
Behavioral Sciences (IRB-SBS) reviews and oversees non-medical, behavioral research studies. The IRB for Health
Sciences Research (IRB-HSR) oversees all other studies involving human subjects, representing the majority of
human use protocols performed by the School of Medicine. Investigators who are new to clinical research or who
wish to perform unfamiliar studies should contact the IRB-HSR prior to submitting a protocol. The Clinical Trials
Office can facilitate the conduct of clinical studies by assisting with budget and proposal preparation, study
coordination and management, and regulatory functions such as quality assurance/quality control.
Animals in research. The Institutional Animal Care and Use Committee (IACUC) reviews and oversees the use of
animals in research and teaching at the University. The IACUC provides training in handling research animals,
insures that individuals using research animals participate in the occupational health and safety program, and
conducts inspections of animal use facilities. The IACUC has specific protocol submission deadlines. The Center for
Comparative Medicine operates UVA vivaria and provides veterinary support. Current per diem rates are listed on
the CCM web site.
Recombinant DNA and pathogens. The Institutional Biosafety Committee (IBC) oversees the use of recombinant
DNA, organisms requiring at least Biosafety Level 2 conditions, and of human specimens. The IBC also inspects
laboratories that have registered for one or more of these activities.
Biohazardous materials (bloodborne pathogens, radiation, hazardous chemicals, shipping biological materials,
etc.). The Office of Environmental Health and Safety maintains comprehensive programs for the management of
potential hazards that may be encountered during research activities. Their web site includes current health and
safety policies and information on ordering radioactive materials. Click here for UVA training requirements,
programs, and on-line training.
Research
General information
Emergency information
Other offices supporting
research
Research resources and
collaborators
Research core facilities
For new faculty
For administrators
For students and postdocs
Intellectual property and
entrepreneurial activities
SOM review of proposed
consulting agreements
Forms and documents
Developing and submitting
a proposal
Where's that e-system?
FAQs
Links
Contact us
Home
Instructions for completing
CITI Conflict of Interest
Modules
Compliance and Training
Medicine PeoplePeople Search site U.Va. U.Va.